
Every element on the Periodic Table has its name listed along with a shorter symbol that we use to refer to it when writing out chemical formulas. Just as important as the atomic number of an element is the name we call it and the symbols we use to refer to it. Sometimes various elements and substances get mixed together to make up things like air or rocks. When elements combine, they create new substances such as water or steel. Modern Periodic Tables recognize 118 elements.Ītoms of alike or different elements will combine to form molecules, a group of atoms, in chemical reactions. Using technology, however, scientists have been able to create additional elements that do not naturally occur. Roughly 90 of these elements occur naturally on Earth. You won’t see these variations listed on the Periodic Table, but the atomic mass that you see for various elements hints at these isotopes because it is an average of the most common masses for that element in nature.Īn element of any substance cannot be broken down into simpler substances by ordinary chemical processes. Atoms of the same element that have a different amount of neutrons than what is most common in nature are called isotopes. Usually, the number of neutrons in an element’s atom will be the same, but sometimes there are differences. Atoms of the same element will have the same number of protons in their nucleus. That means all of the elements that are alike will have atoms with the same structure and characteristics. On the next page, we will discuss in detail what each of these things mean.Īn element is a pure substance that is made up of only one kind of atom. These things help you tell the different elements apart from each other. 
Note that the periodic table above has a slightly different key, and the atomic mass is on top, next to the atomic number. This diagram shows you the key details you will need to identify when looking at an element on the Periodic Table: its atomic number, chemical symbol, chemical name, atomic mass, and some other details you won’t need to know immediately. You will see that some periodic tables will give you much more information, but, for now, we will stick to the basics. The information in this box is a little simplified. It presents an enlarged version of one of the boxes you will see on the Periodic Table. When you learn more about how the Periodic Table is organized, though, you will see that it shows you a lot of useful information about elements. At first, it seems like there’s no telling what it could all mean. It’s filled with a bunch of numbers and symbols. Gtsave(gt_final, filename = "periodic-table.Reading the Periodic Table can seem to be very confusing at first. # change footnote symbol (can't choose specifically those used on Wikipedia) Locations = cells_column_labels(columns = "12") 
Locations = cells_column_labels(columns = "3")įootnote = "Some authors treat Zn, Cd and Hg as transition metals.", (b) The last two members of the group are known as transition metals.", Table_ = "#FFFFFF",Ĭolumn_ = "#FFFFFF"įootnote = "(a) Whether group 3 is composed of -La-Ac or -Lu-Lr is under review by the IUPAC. # remove borders for non-cells places by putting them in white as "style = NULL" doesn't work # create markdown instruction to make numbers and abbreviations on separate lines So here it is! Nothing complicated, a bit tedious to make, but a nice introduction to this package. For reference, this is the table I tried to replicate. I stumbled upon a periodic table of elements, and thought it would be a good exercise to reproduce it, as I am not familiar with the gt package. 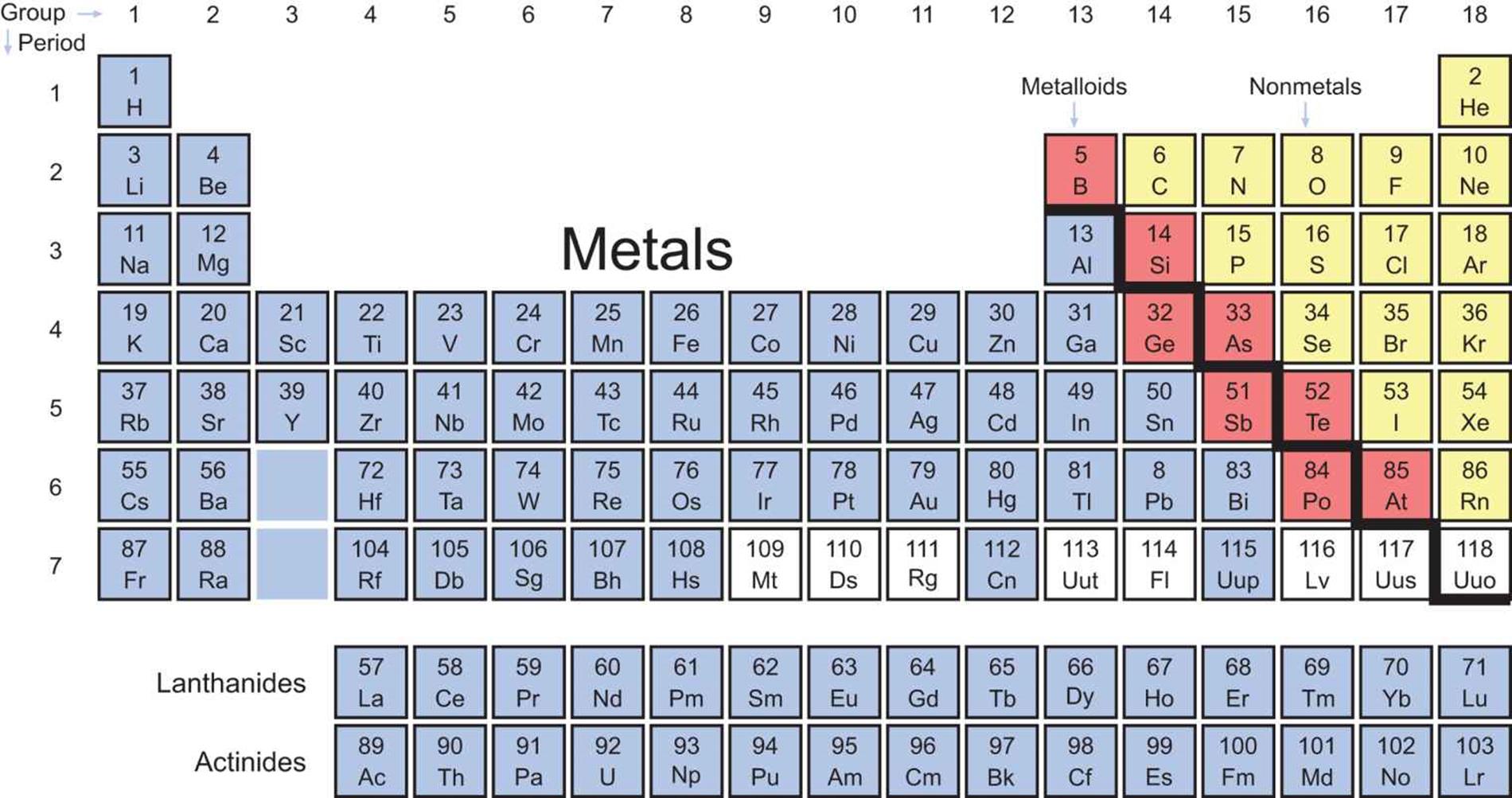
So I was thinking I would not participate to this, but I tried to search "tables stats" online to see if something inspired me. Contrarily to the Shiny contest, I didn't have any idea for this contest as I rarely see beautiful tables that I would like to reproduce, and I don't need to create particularly complicated tables in my work.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
